The life sciences and global MedTech regulatory landscape is highly complex, with rapidly evolving regulations and varying regional requirements impacting time-to-market and the overall success of drug discovery and development.
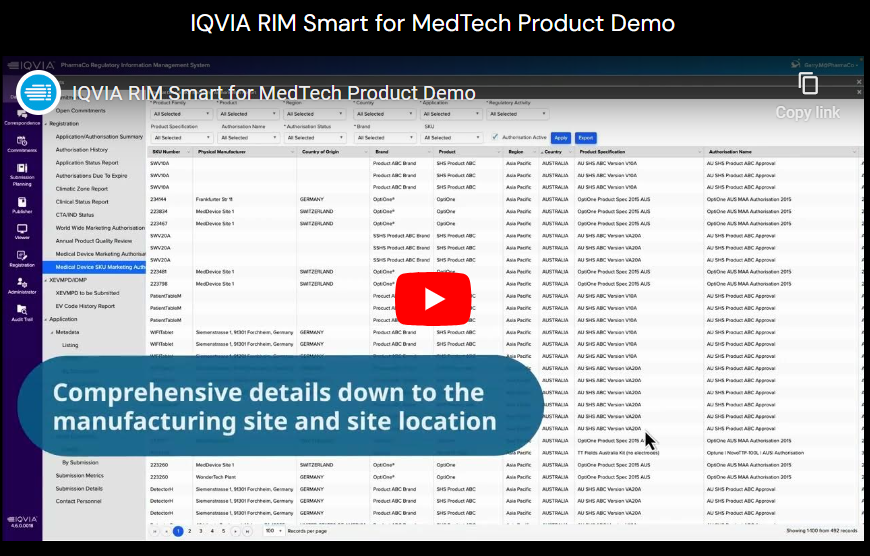
IQVIA RIM Smart is a new cloud-based technology, integrating regulatory, safety and quality data, to support product regulatory lifecycles. Explore the intelligent features of its configurable dashboard, and discover the seamless support it offers to global teams, including:
- Task management for tracking assignments, managing commitments, and monitoring correspondence
- Reporting and tracking capabilities for extracting information on manufacturing sites, locations, and renewal planning
- Submission viewer functionality across various countries, ensuring compliance with local regulations
- Integration with QMS platforms, providing a single source for regulatory intelligence
- Cloud-based technology enabling global team support and scalability